Partners
Development process
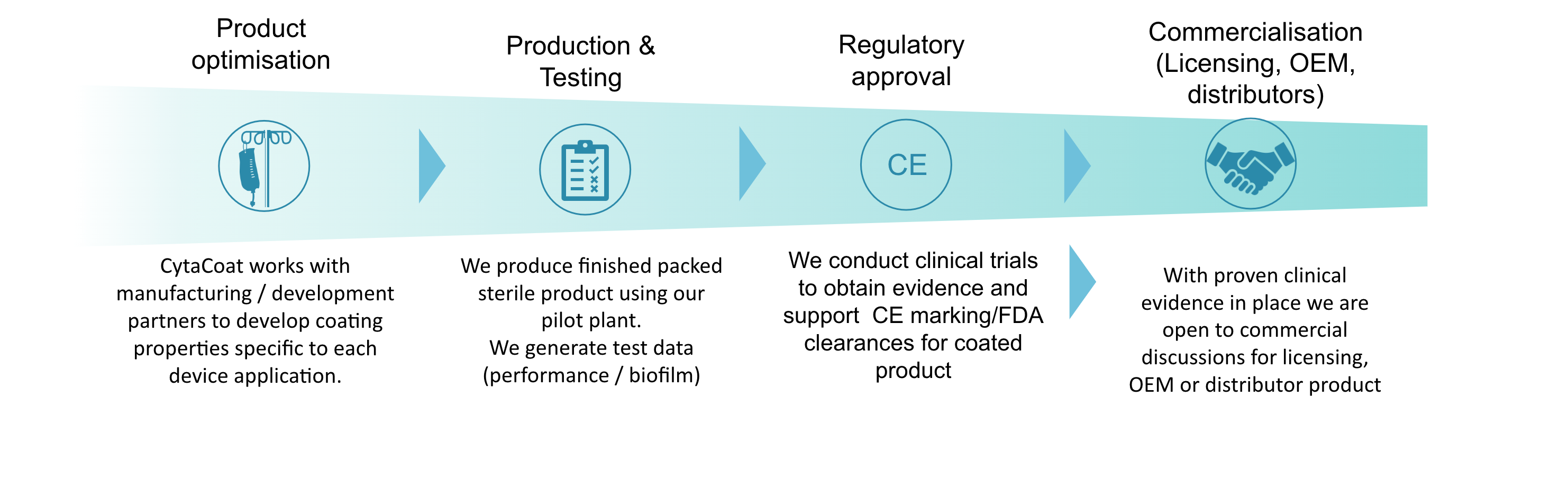
The core of CytaCoat’s business is developing and marketing new coatings for medical devices. We identify a need in the market and develop a coating to fit that device. To do this, we form manufacturing and development partnerships to first develop, produce, and test a coated device then undertake clinical trials to provide evidence for the effectiveness of the coated product in each indication.
CytaCoat is currently active in the urinary catheter and wound care market segments, developing clinical evidence that will support bringing product to market.
CytaCoat routes to commercialisation
CytaCoat is targeting multiple routes to market. We are building the capabilities to produce finished packed sterile product via collaboration with our manufacturing partners and this product will be made available as Original Equipment Manufacturer (OEM) product to medical device companies and also to distributors. Additionally, our technology will be brought to market via licensing to medical device companies for them to coat their products and take them to market.
CytaCoat AB, Maria Aspmans gata 44A, 171 64 Solna SWEDEN | E-mail info@cytacoat.se
Design LIVE Reklambyrå
